Did you know?
The Capra Science Immunization program™ is optimized for high antibody yields and low background.
Monoclonal antibody production
Mouse monoclonal antibody development is generation of new hybridomas expressing antibodies to your antigen of interest. This procedure includes the following steps:
I. Antigen generation (if requested)
The antigen is typically a peptide or a protein.
Ca. 2mg of the antigen is required for the mouse immunization program.
Peptides should at least hold 95% purity.
Shorter peptides will be conjugated to protein carrier (KLH or other).
The antigen is typically a peptide or a protein.
Ca. 2mg of the antigen is required for the mouse immunization program.
Peptides should at least hold 95% purity.
Shorter peptides will be conjugated to protein carrier (KLH or other).
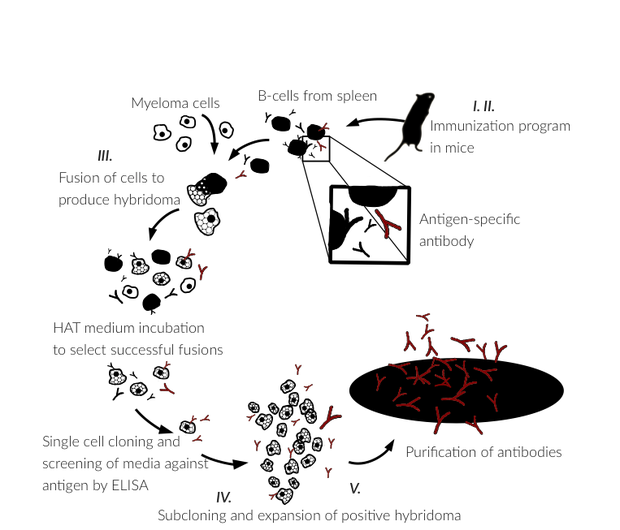
II. 13-week Immunization program
a) Immunization program of four (4) BALB/c mice
b) Immunization scheme includes 1 immunization followed by 3 boosts/mouse
c) The test bleed sample collected at week 13
d) Evaluation of immune response by indirect ELISA of antiserum samples
e) Project report and delivery of antiserum samples, if requested.
a) Immunization program of four (4) BALB/c mice
b) Immunization scheme includes 1 immunization followed by 3 boosts/mouse
c) The test bleed sample collected at week 13
d) Evaluation of immune response by indirect ELISA of antiserum samples
e) Project report and delivery of antiserum samples, if requested.
III. Cell fusion and hybridoma generation
a) Selected mouse is boosted (more than one mouse can be selected)
b) Sacrifice of mouse/mice and spleen cells are extracted
c) Fusion is made with HAT-sensitive myeloma cells and B-cells from spleen
d) Single cell cloning and HAT-incubation
e) Sceening of hybridoma clones by ELISA to select positive clones
f) Cell supernatants from positive clones are collected
g) Project report and delivery of cell supernatants for your evaluation.
Custom option: Cross-reactivity exclusion screening by ELISA. Positive clones can be screened against several molecules.
a) Selected mouse is boosted (more than one mouse can be selected)
b) Sacrifice of mouse/mice and spleen cells are extracted
c) Fusion is made with HAT-sensitive myeloma cells and B-cells from spleen
d) Single cell cloning and HAT-incubation
e) Sceening of hybridoma clones by ELISA to select positive clones
f) Cell supernatants from positive clones are collected
g) Project report and delivery of cell supernatants for your evaluation.
Custom option: Cross-reactivity exclusion screening by ELISA. Positive clones can be screened against several molecules.
IV. Sub cloning
a) The hybridomas of you choice are sub cloned
b) Sub cloned hybridomas are expanded
c) Cell supernatants from several sub clones/hybridoma are collected
d) Project Report and delivery of cell supernatants for your evaluation
a) The hybridomas of you choice are sub cloned
b) Sub cloned hybridomas are expanded
c) Cell supernatants from several sub clones/hybridoma are collected
d) Project Report and delivery of cell supernatants for your evaluation
V. Expansion and Monoclonal Antibody Production
a) The sub cloned hybridoma of your choice is expanded to a larger Culture
b) Media is affinity purified through a protein G column to extract pure IgG
c) Buffer and concentration adjusted to your request
d) Delivery of minimum 5mg antibody with final Project Report
Custom option: Your hybridoma can be dry-ice shipped to you at your convenience. You are also able to securely store the hybridoma at Capra Science for a minor monthly fee.
a) The sub cloned hybridoma of your choice is expanded to a larger Culture
b) Media is affinity purified through a protein G column to extract pure IgG
c) Buffer and concentration adjusted to your request
d) Delivery of minimum 5mg antibody with final Project Report
Custom option: Your hybridoma can be dry-ice shipped to you at your convenience. You are also able to securely store the hybridoma at Capra Science for a minor monthly fee.
Each step of the process is evaluated before you decide whether or not you want to commit to the next phase of development.
Our work is based on a fee-for-service model. Hence, there will be no additional payments such as milestones, royalties or licence costs. We are open to signing CDA/MTA documents prior to project discussions or receiving your material.
This technology of monoclonal antibody generation is commonly used for developing products that are in clinical diagnostics. Moreover, there are several approved drugs that have resulted from antibody humanization of developed mouse monoclonal antibodies using this technique. If you need to humanize your antibody at a later stage, we can assist you.
Our work is based on a fee-for-service model. Hence, there will be no additional payments such as milestones, royalties or licence costs. We are open to signing CDA/MTA documents prior to project discussions or receiving your material.
This technology of monoclonal antibody generation is commonly used for developing products that are in clinical diagnostics. Moreover, there are several approved drugs that have resulted from antibody humanization of developed mouse monoclonal antibodies using this technique. If you need to humanize your antibody at a later stage, we can assist you.